- Blog
- Diablo 2 maphack 1-13 d download
- Epson printer help
- Descargar bloques autocad 2010
- Daisy co2 200 seal kit
- Vienna ensemble pro 7 trial
- Solidworks 2012 simulation tutorial
- Verizon mifi 4510l manual pdf
- Lamin-b association with dna repair factors
- Doordash driver app
- Caliper transcad
- Farm games to play outside
- What is ntfs for mac
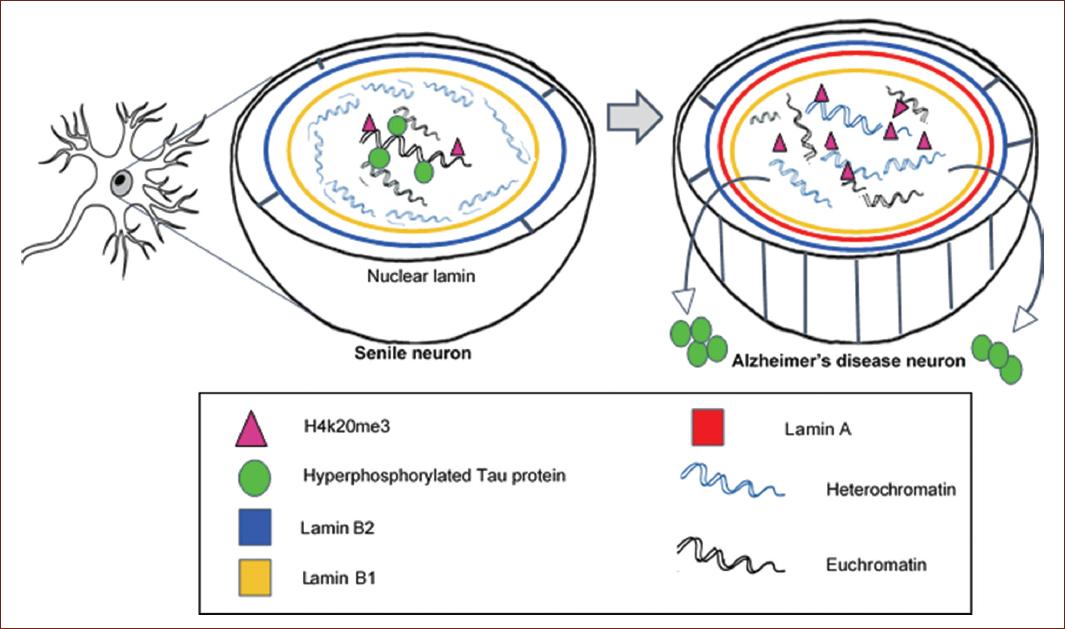
The nuclear lamina is a filamentous mesh network that lines the inner nuclear membrane of metazoan nuclei. Therefore, they not only add a new layer to the study of epigenetic changes in the progeroid syndrome, but also advance our understanding of the disease’s pathology at the cellular level.

Taken together, our results strongly implicate epigenetic deregulation of LADs as an important and previously unrecognized feature of HGPS, which contributes to disease-specific gene expression. Finally, we show that the epigenetic deregulation of LADs is associated with HGPS-specific gene expression changes. Moreover, HGPS patients can be stratified into two different subgroups according to their DNA methylation profiles. Infinium MethylationEPIC BeadChip profiling further reveals that DNA methylation alterations observed in HGPS fibroblasts are similarly enriched in LADs and different from those occurring during healthy aging and Werner syndrome (WS), another premature aging disease. Our ATAC-see/-seq data demonstrate that primary dermal fibroblasts from HGPS patients exhibit chromatin accessibility changes that are enriched in LADs. To do so, we studied chromatin accessibility (Assay for Transposase-accessible Chromatin (ATAC)-see/-seq), DNA methylation profiles (Infinium MethylationEPIC BeadChips), and transcriptomes (RNA-seq) of nine primary HGPS fibroblast cell lines and six additional controls, two parental and four age-matched healthy fibroblast cell lines. Here, we analyze the possibility that epigenetic deregulation of lamina-associated domains (LADs) is involved in the molecular pathology of HGPS.

Cells from affected individuals express a mutant version of the nuclear envelope protein lamin A (termed progerin) and have previously been shown to exhibit prominent histone modification changes. Hutchinson-Gilford progeria syndrome (HGPS) is a progeroid disease characterized by the early onset of age-related phenotypes including arthritis, loss of body fat and hair, and atherosclerosis.
- Blog
- Diablo 2 maphack 1-13 d download
- Epson printer help
- Descargar bloques autocad 2010
- Daisy co2 200 seal kit
- Vienna ensemble pro 7 trial
- Solidworks 2012 simulation tutorial
- Verizon mifi 4510l manual pdf
- Lamin-b association with dna repair factors
- Doordash driver app
- Caliper transcad
- Farm games to play outside
- What is ntfs for mac
